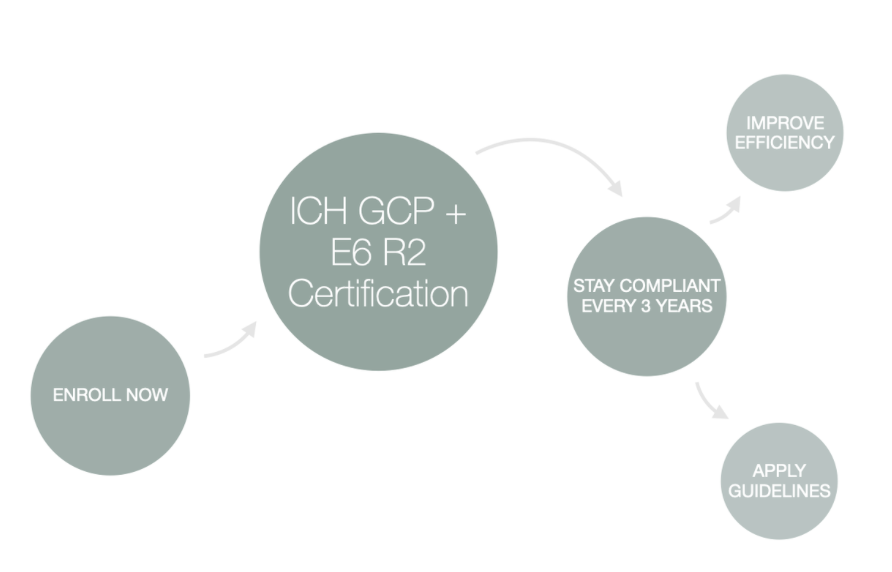
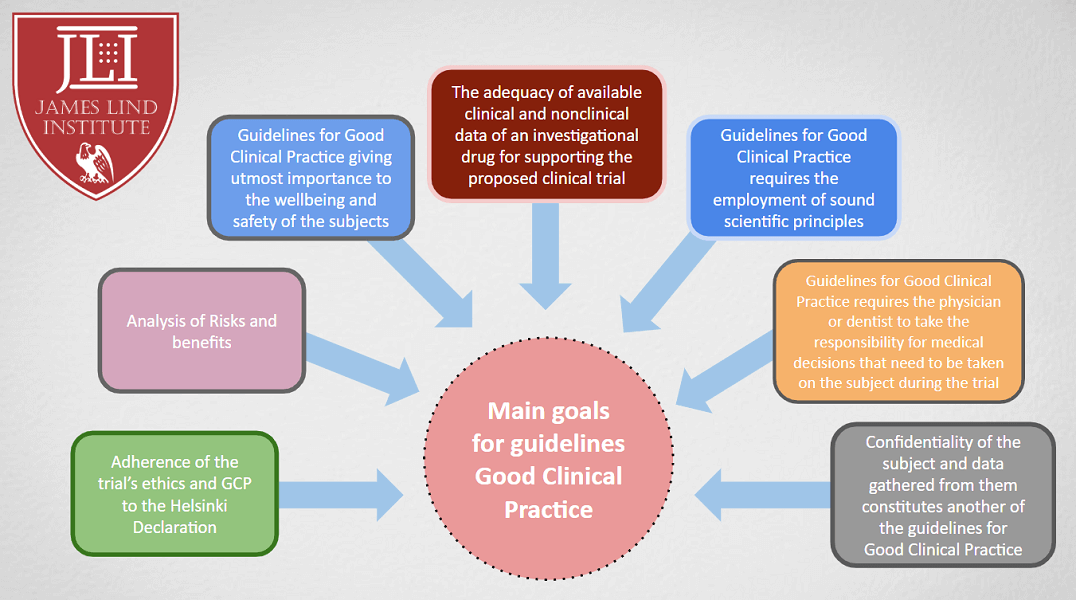
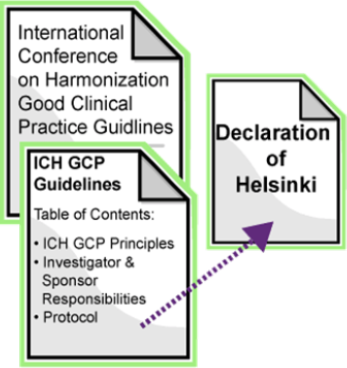
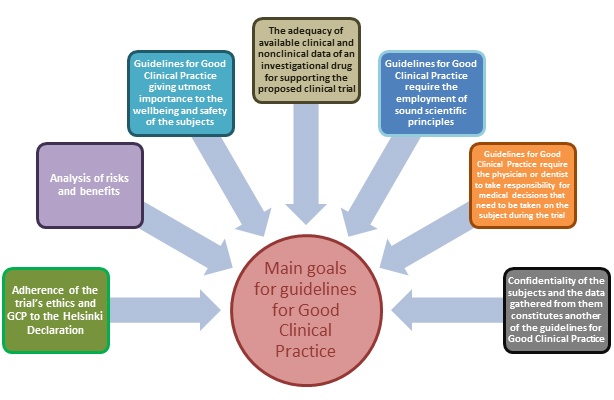
Consorzio per Valutazioni Biologiche e Farmacologiche - CVBF organises the “ICH-Good Clinical Practice (GCP) Training Course”, an e-learning training course aimed at providing a guide for all individuals that are involved in

AO Foundation в Twitter: „The AO's Good Clinical Practice (GCP) course for surgeons takes place November 23-24, in Dübendorf, Switzerland. Register now! https://t.co/uRZ5yXndRU #AOFoundation #AOPEER #research #education https://t.co/gIgbFXwkci“ / Twitter

Clinical Trials Audit Preparation: A Guide for Good Clinical Practice (GCP) Inspections: 9780470248850: Medicine & Health Science Books @ Amazon.com
The Good Clinical Practice (GCP) and the responsibilities of pharma sponsors - Avantyo article in Viata Medicala magazine · News · Avantyo

ICH GCP - 8. Essential documents for the conduct of a clinical trial: ICH E6 (R2) Good clinical practice

REFRESHER: ICH Good Clinical Practice (GCP) E6 (R2) and regulatory requirements for Clinical Trials (Fiona Stanley Hospital) - RETProgram

Good Clinical Practice (GCP) training: Helping you find your next clinical research job | Proclinical Blogs