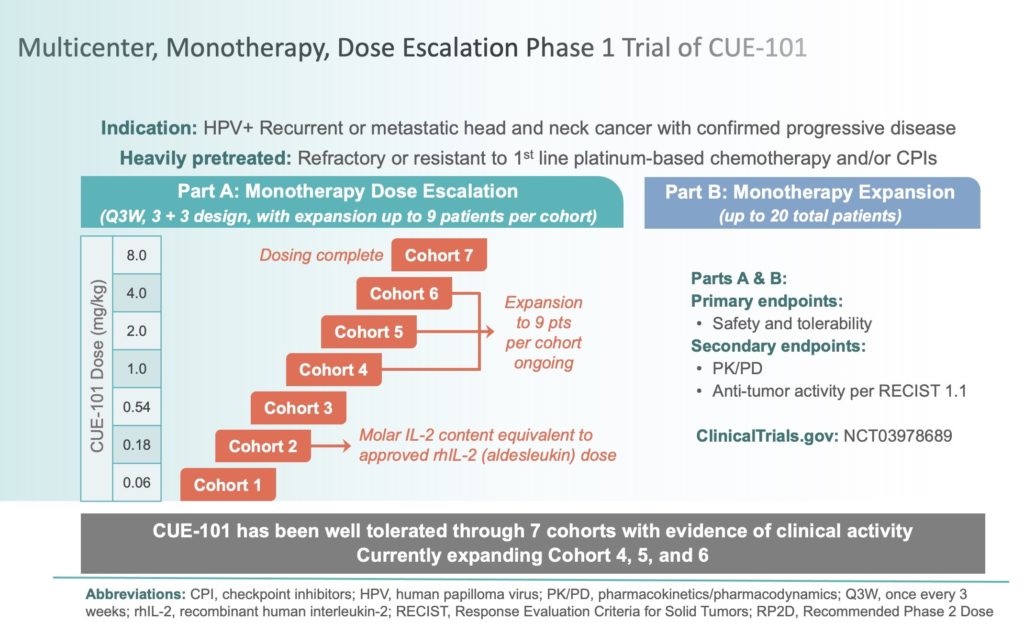
Buy Becoming a Successful Clinical Trial Investigator: A Step by Step Guide for Developing a Clinical Trial Site Book Online at Low Prices in India | Becoming a Successful Clinical Trial Investigator:

Schema of preclinical and clinical pharmacology studies important for... | Download Scientific Diagram

On Biostatistics and Clinical Trials: Serial Blood Sample Timepoints for Comparing Pharmacokinetics Profiles Between Two Different Dose Frequencies

Optimal biological dose: a systematic review in cancer phase I clinical trials | BMC Cancer | Full Text
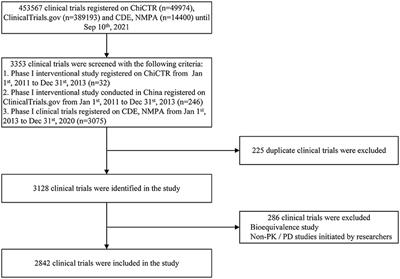
Frontiers | Trends of Phase I Clinical Trials of New Drugs in Mainland China Over the Past 10 Years (2011–2020)

Pharmacokinetic and Statistical Considerations in First-in-Human Clinical Trials | Pharmaceutical Outsourcing - The Journal of Pharmaceutical & Biopharmaceutical Contract Services
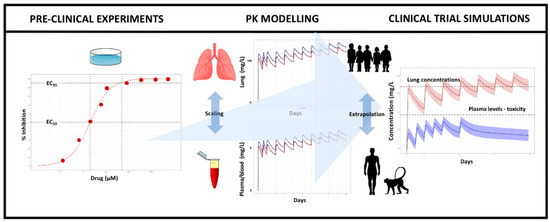
Pharmaceutics | Free Full-Text | Model-Informed Repurposing of Medicines for SARS-CoV-2: Extrapolation of Antiviral Activity and Dose Rationale for Paediatric Patients

Pharmacokinetic and Statistical Considerations in First-in-Human Clinical Trials | Pharmaceutical Outsourcing - The Journal of Pharmaceutical & Biopharmaceutical Contract Services

Current status and future perspective on preclinical pharmacokinetic and pharmacodynamic (PK/PD) analysis: Survey in Japan pharmaceutical manufacturers association (JPMA) - ScienceDirect